The counterpart of commercial carbonate-based electrolytes in LIB is not apt for SIBs due to medium Lewis acidity of Na, less charge density, and more soluble SEI in the electrolyte. Additionally, commercial layered oxide cathodes of LIBs undergo phase changes in NIB counterparts due to diverse chemistry. The water sensitivity of oxide cathodes poses strict storage conditions to prevent CEI instability. Also, nucleophilic attack at deep discharge induced by oxygen anion redox reaction causes the release of CO 2 gas in carbonate-based electrolytes. Due to the catalytic nature of transition metal oxides, the electrolyte can undergo redox reaction, degradation, transition metal dissolution, migration to the anodic side, and dissolution of SEI. On the cathode side, CEI formation at high voltages is mainly impacted by the surface chemistry of the electrode. The presence of huge surface functionality can induce undesired side reactions, electrolyte decomposition, and thicker SEI. These factors influence the degree of solvation/desolvation of Na ions, the distribution gradient of solvent molecules, and the coupling strength of ions. The tunable structural properties of carbon anode depend upon the porosity type, defect density, and surface functional groups. Besides this, surface reactivity and reaction sites of the different carbon materials provide different sodiation behavior. These factors are battery systems, the type of precursor for anode, electrolyte composition, and binder. 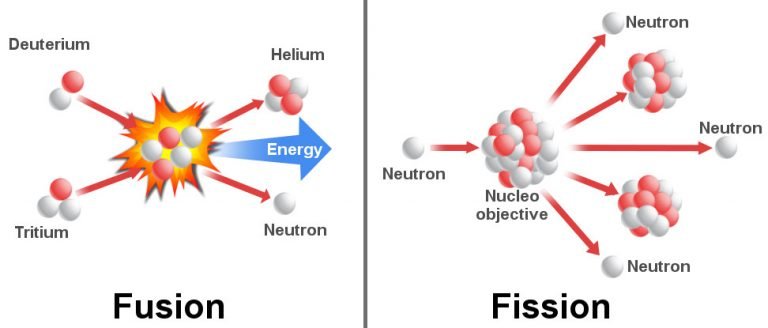
In the most commonly used HC anode materials for SIBs, several factors influence the nature of the SEI. However, SEI in SIBs is more soluble in the electrolyte than its LIB counterpart. SEI should be ionically conducting, impermeable to solvent molecules, electrically insulating, and inert to parasitic reactions. Also, unstable electrolyte composition cannot sustain negative anode potential and forms unstable SEI. Irreversibility of the electrochemical reaction at the interface affects the C.E. The main accountable parameter for the poor performance of SIBs is irreversible Na ion consumption in Solid Electrolyte Interface (SEI) layer formation on the anode and Cathode Electrolyte Interface (CEI) formation on the cathode which consumes most of the first cycle capacity or Na irreversibly and it is known as irreversible capacity loss (ICL) that decreases C.E. Interfaces decide the ionic and electrical conductivity as well as the adhesion of electrode components. Along with the judicial selection of electrode materials, electrolytes, and interfaces are very crucial for safe, high-power, and long-lasting batteries. Transformational research is necessary to fetch SIB from the lab to market at a low cost. However, the far-reaching commercialization of SIBs has been shackled by several challenges such as huge first cycle capacity loss, low columbic efficiency (C.E.), low capacity in a full cell, missing suitable electrolytes for high energy density batteries, etc. Several companies have developed commercial prototypes for SIBs such as Faradion, Natron, Tiamat, Novasis, and CATL. Moreover, the severity of dendrite formation is expected to be less than Li due to the softer nature of Na and conventional LIB fabrication facilities can be directly employed in SIBs. Also, fluorophosphate cathode can provide equivalent energy density (507 Whkg -1) to LiFePO 4 (LFP) counterpart (580 Whkg -1) in LIBs. SIBs are a potential alternative candidate to replace LIBs due to their low cost, sustainability, secure energy supply, and worldwide accessibility.Īdded advantages of SIBs include earth abundance (4 th most abundant element), a low-cost Al current collector that can be used at both the anode and cathode side (Al does not alloy with Na metal and is lighter in weight than Cu current collector which is used in LIBs), safe transportation at 0 V (LIBs are required to transport at 30% SOC), high power, and fast charging,, ]. Although many chemistries are in the trial such as high voltage cathodes and multivalent batteries, ,, , ]. Due to supply constraints, single energy storage technology will not be sufficient to meet the world’s increasing energy demand at a low cost. To decrease the carbon footprint and greenhouse gas emissions, the development of high-performance energy storage technology is essential.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
